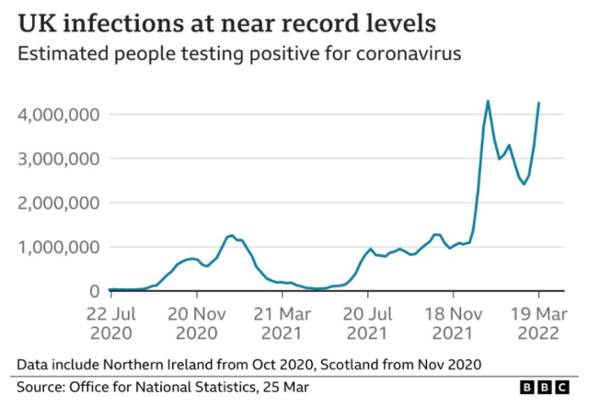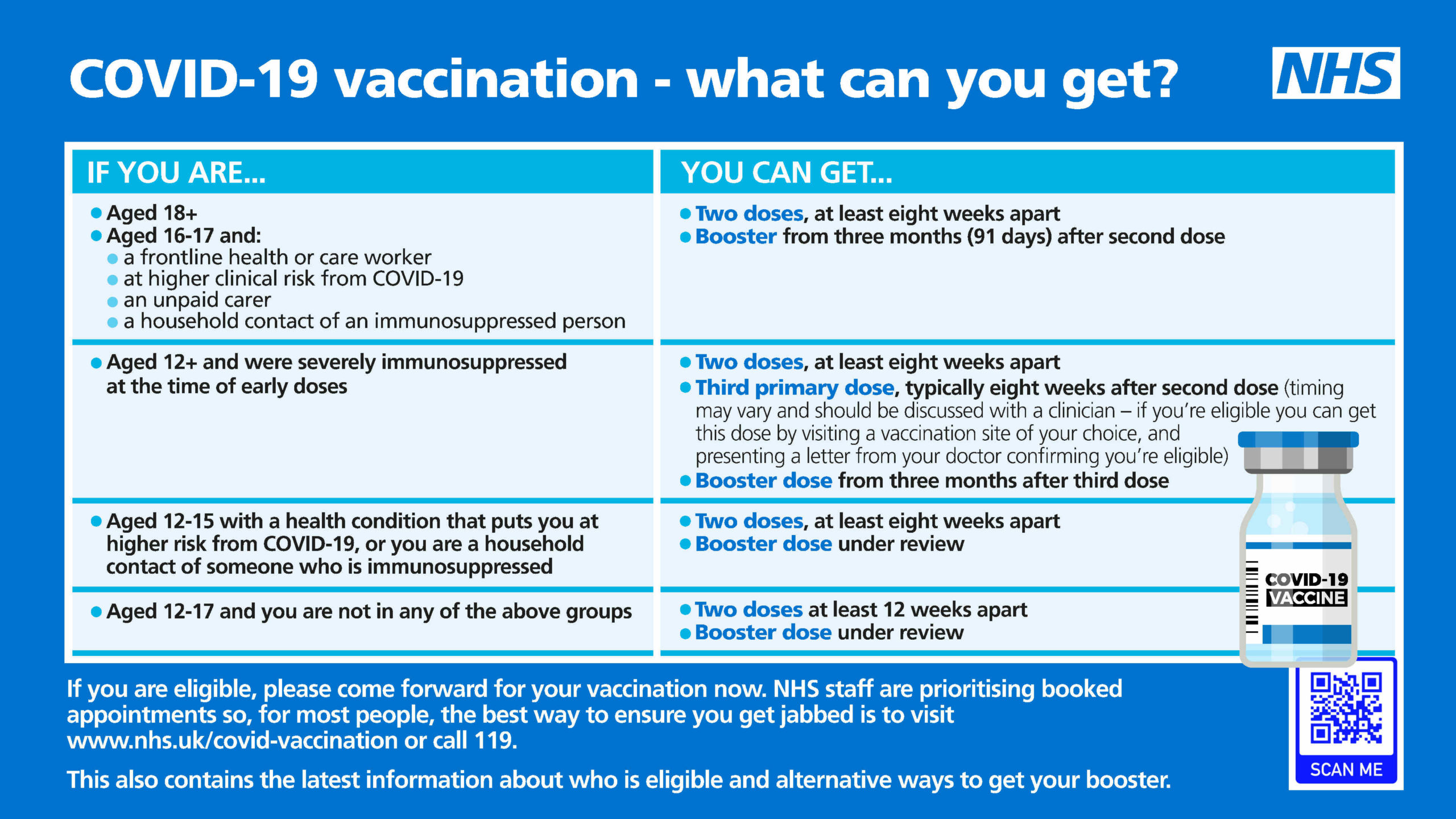
COVID vaccinations: questions and answers about the rollout in North Yorkshire - NHS North Yorkshire CCG
8 in 10 UK citizens willing to donate their COVID-19 booster dose if not needed | Imperial News | Imperial College London

80% of patients admitted to Intensive Care Units in north east London in December not fully vaccinated - NHS North East London

Severe COVID-19 outcomes after full vaccination of primary schedule and initial boosters | The University of Edinburgh